- Blog
- Super mario galaxy 2 dolphin xbox controller
- Change dates in microsoft word calendar template
- Free template business expense report in excel
- Hard signs neck trauma
- Age empires 3 download free full version
- Remo recover 4-0 licence key
- Free newspaper templates for word
- Meri shaadi mein zaroor aana online watch
- Free motion backgrounds for church
- Half life 1 texture glitch
- Color coded periodic table with ion charges
- Ada cad drawings
- Kmspico for office 2016 activator
- Desktop photo calendar 2020
- Meal planner based on macros
- Grateful dead hippie font
- Photo calendar desktop
- Reddit terraria free pc download
- Learning to program with robotc
- Photoshop cracked 2019 mac torrent
- Car air conditioner maintenance checklist
- Family tree for queen victoria
- Jay z and beyonce run video
- Family movies on netflix 2017
- Minecraft forge unnamed installation
- The periodic table color coded
- Professional website service invoice template
- New jersey seashore points map
- 3d comics chaperone episode 3
- Periodic table periodic table with molar mass
- Ssh multiple sessions putty
- Inmate icare packages
- Two person household budget excel
- Instant pot macaroni and cheese with bacon
- Flipnote studio 3d download link
- Cura 15-04-6 download dmg
- Vlc media player record pc audio output
- Siberian mouse masha naked gif
- Recipe for oatmeal raisin cookies
- Track ip address of facebook account
- Farming simulator 19 multiplayer cars
- Happy wheels full game free play
- The railway journey schivelbusch jsto
- Couple bangs babysitter
- Blog
- Super mario galaxy 2 dolphin xbox controller
- Change dates in microsoft word calendar template
- Free template business expense report in excel
- Hard signs neck trauma
- Age empires 3 download free full version
- Remo recover 4-0 licence key
- Free newspaper templates for word
- Meri shaadi mein zaroor aana online watch
- Free motion backgrounds for church
- Half life 1 texture glitch
- Color coded periodic table with ion charges
- Ada cad drawings
- Kmspico for office 2016 activator
- Desktop photo calendar 2020
- Meal planner based on macros
- Grateful dead hippie font
- Photo calendar desktop
- Reddit terraria free pc download
- Learning to program with robotc
- Photoshop cracked 2019 mac torrent
- Car air conditioner maintenance checklist
- Family tree for queen victoria
- Jay z and beyonce run video
- Family movies on netflix 2017
- Minecraft forge unnamed installation
- The periodic table color coded
- Professional website service invoice template
- New jersey seashore points map
- 3d comics chaperone episode 3
- Periodic table periodic table with molar mass
- Ssh multiple sessions putty
- Inmate icare packages
- Two person household budget excel
- Instant pot macaroni and cheese with bacon
- Flipnote studio 3d download link
- Cura 15-04-6 download dmg
- Vlc media player record pc audio output
- Siberian mouse masha naked gif
- Recipe for oatmeal raisin cookies
- Track ip address of facebook account
- Farming simulator 19 multiplayer cars
- Happy wheels full game free play
- The railway journey schivelbusch jsto
- Couple bangs babysitter
One way chemists describe matter is to assign different kinds of properties to different categories. Part of understanding matter is being able to describe it. Chemistry lies more or less in the middle, which emphasizes its importance to many branches of science. Figure 2.1 shows how many of the individual fields of science are related.įigure 2.1: The Relationships Between Some of the Major Branches of Science. Similarly, geology and chemistry overlap in the field called geochemistry. For example, some biologists and chemists work in both fields so much that their work is called biochemistry. Mathematics and Physics are the languages of science, and we will use them to communicate some of the ideas of chemistry.Īlthough we divide science into different fields, there is much overlap among them. Thus, chemistry is the study of matter, biology is the study of living things, and geology is the study of rocks and the earth. Because the physical universe is so vast, there are many different branches of science (Figure 2.1). is the process by which we learn about the natural universe by observing, testing, and then generating models that explain our observations. Science is the process by which we learn about the natural universe by observing, testing, and then generating models that explain our observations. This includes advances in medicine, communication, transportation, building infrastructure, food science and agriculture, and nearly every other technical field that you can imagine.Ĭhemistry is one branch of science. From the color that makes a rose so red to the gasoline that fills our cars and the silicon chips that power our computers and cell phones…Chemistry is everywhere! Understanding how chemical molecules form and interact to create complex structures enables us to harness the power of chemistry and use it, just like a toolbox, to create many of the modern advances that we see today. Section 2.1: Chemistry and Matter What is Chemistry?Įverything around us is made up of chemicals.

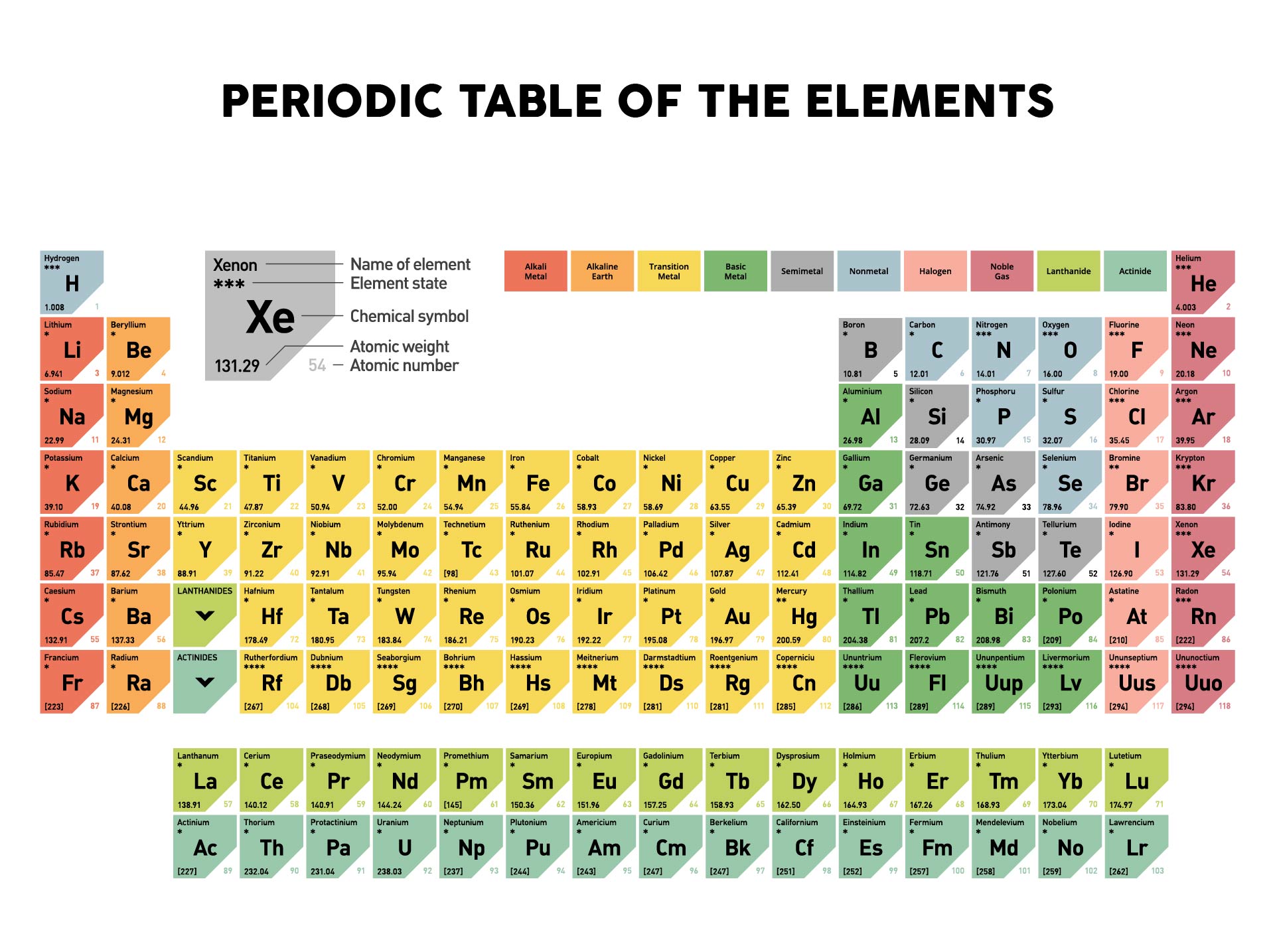
One mole of a substance has the same mass in grams that one atom or molecule has in atomic mass units.Chapter 2: Atoms and the Periodic Table Section 2.1: Chemistry and Matter What is Chemistry? Physical and Chemical Properties Elements and Compounds Mixtures States of Matter Reactions in Chemistry Section 2.2: How Scientists Study Chemistry The Scientific Method 2.3 Atomic Theory with Historical Perspectives 2.4 Introduction to Elements and the Periodic Table 2.5 Dmitri Mendeleev and the development of the periodic table 2.6 Families of the Periodic Table 2.7 Defining the Atom Basic Atomic Structure – electrons, neutrons, and protons 2.8 Atomic Number – Protons Determine the Identity of an Element 2.9 Atomic Mass, Isotopes, and Molar Mass 2.10 Periodic Table Trends Atomic Size Electronegativity Ionization Energy Metallic and Nonmetallic Character 2.11 Chapter Summary and Homework 2.12 References
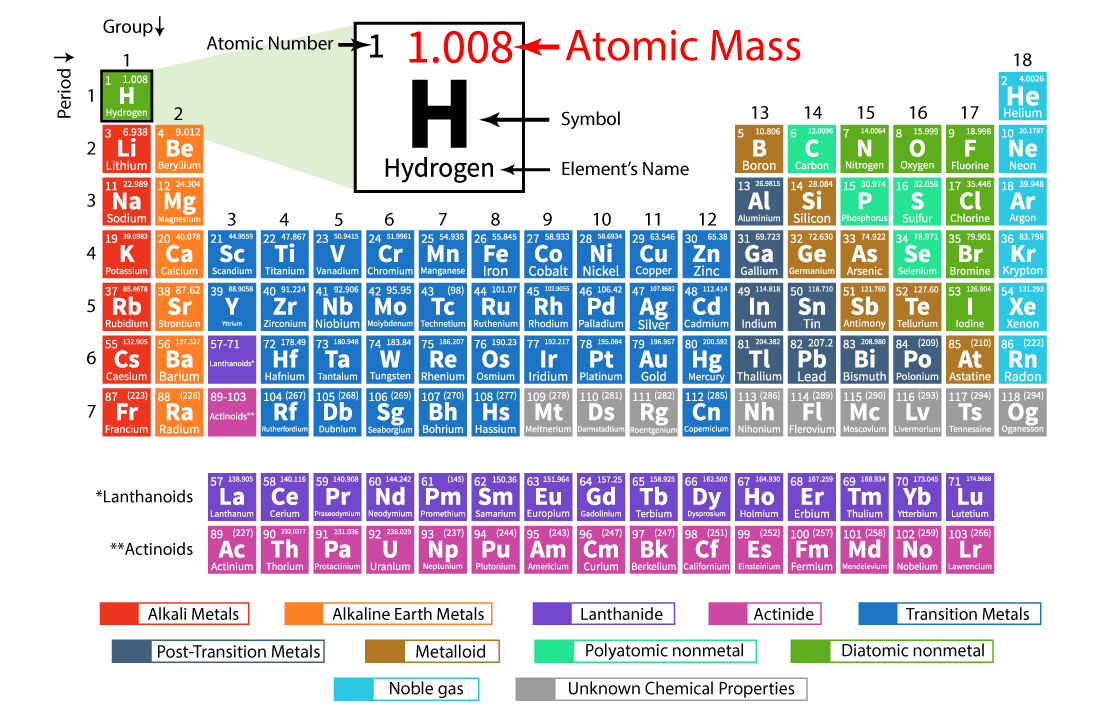
The numbers in the periodic table that we identified as the atomic masses of the atoms not only tell us the mass of one atom in u but also tell us the mass of 1 mol of atoms in grams. One mole of a substance has the same mass in grams that one atom or molecule has in atomic mass units. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of Na atoms has an approximate mass of 23 grams. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of H atoms has a mass of approximately 1 gram. The number in a mole, Avogadro’s number, is related to the relative sizes of the atomic mass unit and gram mass units.
